Pfizer Recalls Birth Control Pills
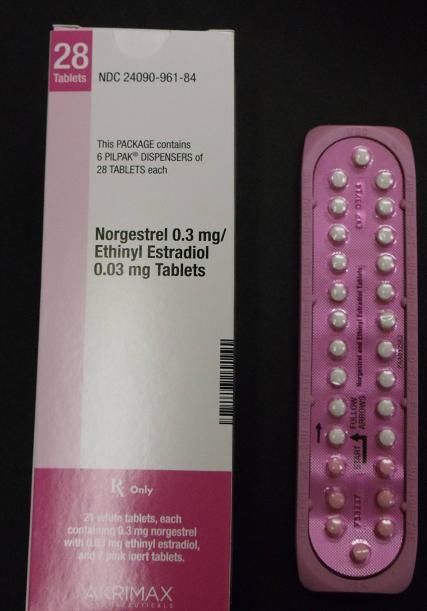
Pfizer Inc. announced today (Jan. 31) that it has voluntarily recalled 14 lots of the birth control pills Lo/Ovral-28, and 14 lots of generic pills labeled Norgestrel and Ethinyl Estradiol tablets.
The recall came after an investigation by Pfizer found that some packs may contain an inexact count of inert or active ingredient tablets, and that the tablets may be out of sequence. The cause was identified and corrected immediately, according to a statement from the company.
As a result of this packaging error, women taking these pills may be without adequate contraception, and at risk for unintended pregnancy, the company said. These packaging defects do not pose any immediate health risks, however, consumers taking pills from the affected lots should begin using a non-hormonal form of contraception immediately. Patients who have the affected product (lot numbers are provided below) should notify their physician and return the product to the pharmacy.
These products are oral contraceptives, indicated for the prevention of pregnancy. These tablets were manufactured and packaged by Pfizer and labeled under the Akrimax Pharmaceuticals brand. This products were distributed to warehouses, clinics and retail pharmacies nationwide, according to the company.
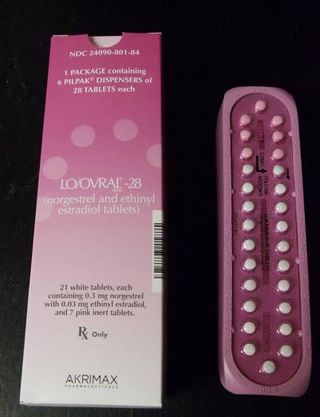
These products are packaged in blister packs, containing 21 tablets of active ingredients and seven tablets of inert ingredients. Correct dosing of this product is important in avoiding an unplanned pregnancy. Correctly packaged blister packs are pictured here.
The company said that any adverse events that may be related to the use of these products should be reported to Akrimax Medical Information at 1-877-509-3935 (8 a.m. to 7p.m. Monday to Friday CST) or to FDA's Med Watch Program either online, by regular mail or by fax.
Online: www.fda.gov/medwatch/report.htm Regular Mail: Use postage-paid, pre-addressed Form FDA 3500 available at: www.fda.gov/MedWatch/getforms.htm. Mail to the address on the pre-addressed form. Fax: 1-800-FDA-0178 This recall is being conducted with the knowledge of the U.S. Food and Drug Administration. Lot numbers of affected packs of Lo/Ovral®-28 (norgestrel and ethinyl estradiol) Tablets and Norgestrel and Ethinyl Estradiol Tablets (generic) follow on the table below:
Sign up for the Live Science daily newsletter now
Get the world’s most fascinating discoveries delivered straight to your inbox.