Can Bodybuilding Hold a Key to Aging?

This article was originally published on The Conversation. The publication contributed this article to Live Science's Expert Voices: Op-Ed & Insights.
People have used bodybuilding to improve their physical performance for at least 1,500 years. The first recorded example was the sixth-century wrestler, Milo of Croton, in southern Italy. Milo reportedly carried out his daily exercises with a calf on his back. As the calf grew into a full sized ox, so too did Milo’s legendary strength.

In the 1940s this concept of what has come to be known as progressive resistance exercise was developed more scientifically by US Army physician Thomas DeLorme to assist the recovery of injured service men and polio sufferers. It is a simple idea: lift a weight you can manage for a set routine and when after numerous training sessions it becomes too light, increase the weight. You repeat the process over and over and it leads to substantially improved strength. This is because the muscle adapts by growing to deal with heavier loads. DeLorme applied these principles to his own physical training and is said to have been formidably strong.
Our little protein factories
Thanks to these insights, in modern-day bodybuilding we now have a very good idea of how to improve muscle mass and strength. Muscle growth with progressive resistance exercise is a good example of how adaptable our skeletal muscle is. Part of the mechanism by which muscle grows is through a process called protein synthesis. Muscle grows by increasing the rate at which proteins are made, since muscle is made from protein.

Protein synthesis also responds to eating dietary protein. And when protein is consumed following resistance exercise, muscle production is enhanced even further. What’s more, muscles that have been subjected to resistance exercises increase their protein synthesis rates more than non-exercised muscles after each meal for at least the next 48 hours.
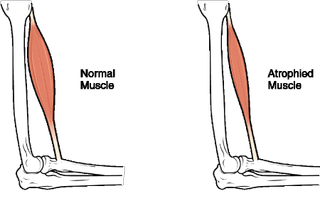
Not only is this insight useful for bodybuilders, it also tells us something useful about muscle wasting. We know that slow wasting conditions such as sarcopenia, where muscle mass progressively diminishes with age, are due to the impaired ability of muscle to increase protein production in response to feeding or loading. In effect, the muscle’s protein-building machinery becomes resistant to growth stimuli. This causes the protein, and hence the muscle, to be lost, which leads to reduced muscle function.
This is important because muscle loss with age is associated with impaired physical function and loss of independence. This is particularly true if it is combined with the muscle wasting that people experience if they are immobilised after injuries or surgery. For example, around 50% of women over the age of 65 who break a hip never walk again. The cause? Loss of muscle mass associated with the injury and the ensuing bed rest.
Sign up for the Live Science daily newsletter now
Get the world’s most fascinating discoveries delivered straight to your inbox.
Sarcopenia is also associated with an increased risk of falling. A review of the cost of treating fall-related injuries in 1999 showed that falls in the over-60s cost the UK government close to a £1bn a year (more than £1.5bn in today’s money). There is therefore a huge public interest in developing safe and effective strategies to prevent muscle wasting.
The research race
We have been aware of this link between muscle wasting and protein synthesis impairment for a long time. You don’t see many old people in the gym lifting weights, but that is exactly what they should be doing to try and offset these effects.
For the vast majority of abstainers, researchers have been looking for potential drug treatments in this area for more than 30 years. And for about the last 15 they have been employing biochemistry techniques in human studies to try and understand the molecular signals that tell the muscle to grow.
These molecular signals act like the foreman on a building site. They read the conditions by sensing whether there is sufficient building materials and manpower to build and whether there has been a change in demand on the existing muscle. They then relay that information into an appropriate growth response to meet the demands placed on the system by initiating or stopping muscle protein synthesis.
The question is which molecules are doing this foreman’s job, which has proved far harder to ascertain than initially might have been hoped. The combined efforts of researchers these past few years have pointed to the likelihood that the culprit is a molecule called p70S6K1. Various teams are now looking at how it potentially leads to more muscle protein being produced, including my own.
One reason why it has taken a long time for anyone to fully explain what happens is because it is hard to measure how this molecule functions in human skeletal muscle. This makes it harder to determine its role in making muscles grow. We have optomised a simple, cost-effective and accurate technique to solve this measuring difficulty. We have already been putting it through human trials, and hope to have a definitive answer in the next five to ten years.
While others take different approaches, the race is on to determine whether p70S6K1 will be a good drug target to treat muscle wasting. After that, it might take another couple of decades to develop commercial drugs. Needless to say, solving the riddle of muscle wasting is not a quick process. But if science finally triumphs here, it will be a great example of how the answers to problems can come from the most unlikely of sources. For now though, the best advice is to take the lead from bodybuilding and undertake a programme of progressive resistance exercise while ensuring you consume adequate amounts of protein.
This article was originally published on The Conversation. Read the original article. Follow all of the Expert Voices issues and debates — and become part of the discussion — on Facebook, Twitter and Google +. The views expressed are those of the author and do not necessarily reflect the views of the publisher. This version of the article was originally published on Live Science.