Cancer Cells Transformed into Harmless Fat in Mouse Study
Imagine if you could turn aggressive cancer cells into harmless fat.
Scientists in Switzerland say they've done just that, in a new study in mice. By taking advantage of the "plasticity," or adaptability, of certain cancer cells during metastasis, the researchers were able to coax breast cancer cells in mice into becoming fat cells.
The scientists accomplished this using a combination of two drugs, both of which are already approved for use in humans by the U.S. Food and Drug Administration (FDA). The treatment didn't convert all of the cancer cells into fat cells, but it did stop the cancer's metastasis, or spread to other parts of the body, the researchers said. [10 Do's and Don'ts to Reduce Your Risk of Cancer]
The work is very preliminary, and it's unclear if the findings will apply to people or to other types of cancers. But because the study used two drugs already approved by the FDA, it "may be possible" that the findings also apply to humans, the researchers wrote in their paper, published today (Jan. 14) in the journal Cancer Cell.
If future studies confirm the new work, the researchers believe that the therapy could be used in combination with conventional chemotherapy "to suppress both primary tumor growth and the formation of deadly metastases," senior study author Gerhard Christofori, a professor at the University of Basel's Department of Biomedicine in Switzerland, said in a statement.
Turning cancer into fat
When cancer cells metastasize, they undergo changes that allow them to "break free" from the initial tumor and spread to another site in the body. In order to do this, the cells temporarily enter a more "immature" state, similar to what's seen in stem cells. In scientific terms, this change is known as an epithelial-mesenchymal transition (EMT).
During EMT, the cancer cells are in a highly plastic, or adaptable, state. This state may offer "a window of opportunity" for therapies to target these cells and force them to transform into a different cell type, the researchers said.
Sign up for the Live Science daily newsletter now
Get the world’s most fascinating discoveries delivered straight to your inbox.
To test this hypothesis, the researchers first created a mouse model of human breast cancer by transplanting human breast cancer cells into the mammary fat pads of female mice.
Then, the researchers treated the mice with two drugs: rosiglitazone, which is used in people to treat type 2 diabetes, and trametinib, an anti-cancer drug that inhibits the growth and spread of cancer cells. (Rosiglitazone belongs to a class of drugs known as thiazolidinediones, which bind to receptors that are found mainly in fat tissue and that play a role in a number of biological processes, including the formation of mature fat cells, according to a 2005 paper on the topic. People with diabetes are given the drug because the receptors that it binds to also help increase sensitivity to the hormone insulin, which is involved in regulating blood sugar levels.)
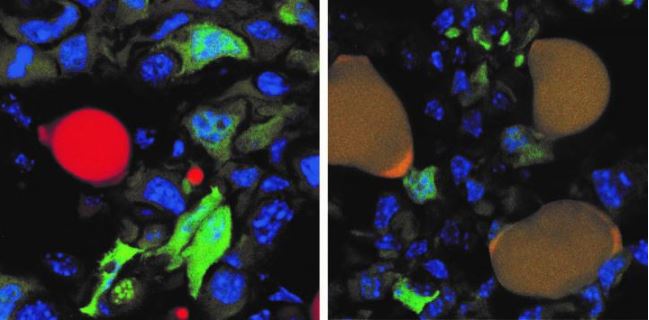
The researchers in the new study found that when mice received this drug combination, the cancer cells that had broken free from the initial tumor (called "invasive cancer" cells) changed into fat cells. The drugs also suppressed the growth of the tumor and prevented further metastasis.
Future research
Targeting cancer cells undergoing EMT "is a new and very elegant idea aimed at turning 'bad' into 'good,'" said Andrei Gudkov, a cancer researcher and senior vice president for research technology and innovation at the Roswell Park Comprehensive Cancer Center in Buffalo, New York, who was not involved in the study. In this case, cancer cells in this adaptable state were forced to change into fat cells that are incapable of further cell divisions, Gudkov noted.
Gudkov agreed that because the study used two FDA-approved drugs, this "greatly facilitates a potential clinical translation" for use in people. However, the design for a study that would unequivocally demonstrate that this drug combination works in cancer patients to prevent metastasis desired "is not obvious," Gudkov said.
It's challenging to discover and test drugs to prevent metastasis, Gudkov said. Such trials need to be long and require a large number of patients. In addition, in this case, the two drugs that would be tested in combination are already FDA approved, and "running generic drugs through lengthy trials rarely happens, in part because of the time and expense involved," Gudkov told Live Science.
The researchers hypothesized that if they forced a "critical mass" of cancer cells undergoing EMT to turn into fat cells, this could reduce the tumor's ability to evade chemotherapy. (EMT is thought to help cancer cells escape chemotherapy, making the cancer cells more adaptable.) In future studies in animals, the researchers said, they plan to test their therapy drug combination with existing chemotherapies; they'll also examine how it affects other types of cancers.
- 7 Odd Things That Raise Your Risk of Cancer (and 1 That Doesn't)
- Ten Things You Didn't Know About Fat
- 5 Ways Your Cells Deal With Stress
Originally published on Live Science.

Rachael is a Live Science contributor, and was a former channel editor and senior writer for Live Science between 2010 and 2022. She has a master's degree in journalism from New York University's Science, Health and Environmental Reporting Program. She also holds a B.S. in molecular biology and an M.S. in biology from the University of California, San Diego. Her work has appeared in Scienceline, The Washington Post and Scientific American.