'I've never seen anything like this': Scientists hijack cancer genes to turn tumors against themselves
Scientists overcame cancer drug resistance in a new proof-of-concept study.
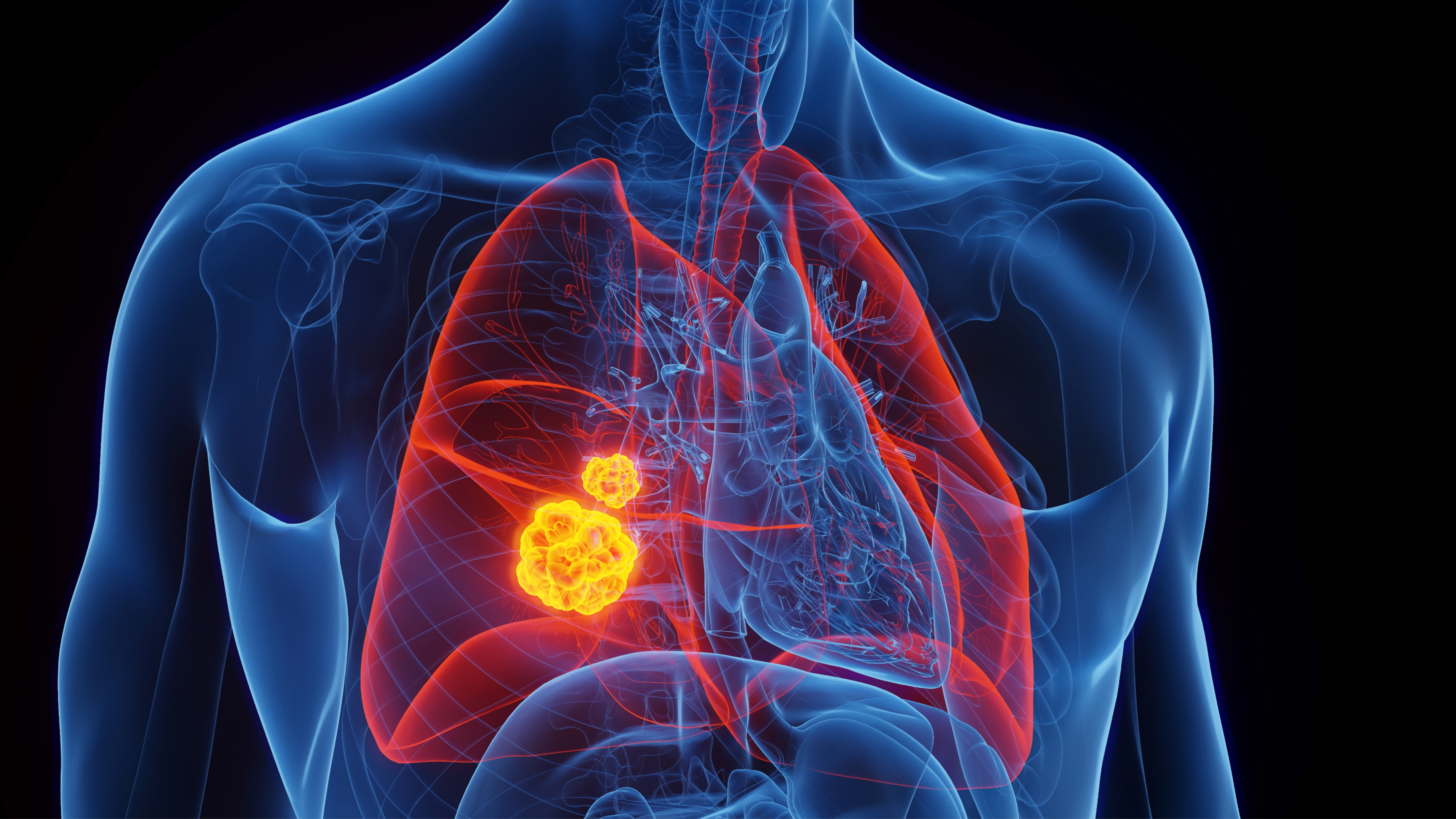
Cancer can sometimes thwart drugs designed to treat it — but now, scientists have developed a way to turn tumor cells against their neighbors, forcing the cancer to cooperate with treatment.
A cancer treatment's success hinges on its capacity to damage cancer cells enough to kill them or stop them from growing. However, some cancer cells can change their molecular makeup to either mitigate or cancel out the therapy's effect.
Now, in a proof-of-concept study, scientists have unveiled a new way of overcoming this cancer drug resistance: hacking the evolution of cancer cells and tagging them with a target that makes them more vulnerable to therapies. The researchers published their findings Thursday (July 4) in the journal Nature Biotechnology.
"There's all this time and effort and energy and money and heartbreaks put into finding drugs that are going to be effective against the next version of the tumor," lead study author Scott Leighow, a bioengineer at Pennsylvania State University, told Live Science. But "no matter how good they are, they're not durable in the long term."
The new approach could combat existing anticancer drug resistance before it becomes insurmountable.
Cancer drug resistance can develop in many ways. For instance, cancer cells can molecularly inactivate a drug or flip internal switches to cheat their own deaths. To try and get around this, doctors can treat patients with combinations of drugs that use different attacks against the tumors. But the approach has limitations.
"The challenge with a lot of these advanced tumors is that we just don't have a lot of good targets to make drugs against," senior study author Justin Pritchard, a biomedical engineer at Pennsylvania State University, told Live Science. "There's not always a second drug that works as well [and] through a totally independent mechanism of action."
Sign up for the Live Science daily newsletter now
Get the world’s most fascinating discoveries delivered straight to your inbox.
Related: The 10 deadliest cancers, and why there's no cure
To tackle the problem from a new angle, Pritchard and his team devised an approach to edit cancer cells, delivering two new "switches" into them. The first switch enables modified cells to outgrow the rest of the cancer cell population. Then, the second switch allows these cells to unleash a toxic drug onto the remaining tumor.
To test the concept, the scientists inserted two "suicide genes" into cancer cells in lab dishes. One gene controls the activation of a protein called epidermal growth factor receptor (EGFR) with the help of a cancer drug called erlotinib.
Normally, erlotinib would stop EGFR proteins from being activated and thus prevent cancer cells from proliferating uncontrollably. But the "suicide gene" allows the scientists to reverse the drug's usual action, making cells resistant to the drug on purpose. This enables them to switch the proliferation of these cells on and off.
For this experiment, the scientists focused on non-small cell lung cancer (NSCLC), the most common type of lung cancer. Most NSCLC cells develop resistance to erlotinib about a year after treatment, which can lead to a relapse. When the scientists introduced both the suicide gene and erlotinib to NSCLC cells, the modified cells easily outgrew unmodified cells that were inherently resistant to the drug. Once the modified cells became dominant, the scientists stopped administering erlotinib, thus switching the cells' proliferation off.
Once their trap was set, the scientists activated the second suicide gene using a harmless molecule called 5-FC. Cleverly, the second gene codes for an enzyme that helps cells transform 5-FC into a toxin, called 5-FU, that kills cancer. The 5-FU ultimately kills both the modified cancer cells and the cells surrounding them.
The scientists call this approach a "dual switch selection gene drive." They tested it in lab mice and found that, about 20 days into treatment, the modified cancer cells overtook the unmodified cells around them. By day 80, the tumor's volume had shrunk down to zero.
"This is innovative, certainly," said Aaron Goldman, a cancer pharmacologist at the Brigham and Women's Hospital who was not involved in the study. Most cancer therapies that involve gene editing focus on engineering immune cells instead of the cancer cells. "I've not seen anything like this," Goldman told Live Science.
While the dual-switch approach is novel, "it does not address resistance generally," Goldman noted. "It certainly addresses a mechanism of resistance — one of many, many." But the approach could be improved if it was combined with a therapy that thwarts cancer cells' ability to develop resistance in the first place, he said. This would both address the existing resistance and prevent future resistance from developing in additional cells.
The researchers are now testing the dual-switch approach with other cancers and cancer therapies. Pritchard and Leighow are founders of Red Ace Bio, a startup company that is developing the technology.
"We see this as something beyond the treatment of lung cancer," Leighow said. "We see this as a generalizable platform for delivering therapeutic genes in cancer and using those to create therapeutic opportunities that might lead to cures in the future."
Ever wonder why some people build muscle more easily than others or why freckles come out in the sun? Send us your questions about how the human body works to community@livescience.com with the subject line "Health Desk Q," and you may see your question answered on the website!

Kristel is a science writer based in the U.S. with a doctorate in chemistry from the University of New South Wales, Australia. She holds a master's degree in science communication from the University of California, Santa Cruz. Her work has appeared in Drug Discovery News, Science, Eos and Mongabay, among other outlets. She received the 2022 Eric and Wendy Schmidt Awards for Excellence in Science Communications.
China uses 'gravitational slingshots' to save 2 satellites that were stuck in the wrong orbit for 123 days
Scientists spot a 'dark nebula' being torn apart by rowdy infant stars — offering clues about our own solar system's past
Mass graves of Black Union soldiers slaughtered by Confederate guerrillas possibly identified in Kentucky